| Regular soap (left) and magnetic soap (right) with magnet. |
Soap has been around for a long time. In itself soap can be changed by changing the amphiphilic head to cationic, anionic or nonionic moeities and the tale can be changed to a long tail, short tail or a tail with a ring structure for example. All these properties help to change the critical micelle concentration and to aid in the lowering of the surface tension of water. A surface tension device can help you tweak the formulations of the different contents of these different surfactants for use in a variety of application. However, there could be so much more that one can do with surfactants and soaps in general that one can do. Imagine for a second light reactive surfactants (respond to different kinds of light), soaps that respond to different kinds of heat (probably done already), soaps that respond to compression (could make some kind of foam that you can stand ontop of) or even soaps that are magnetic.
The last one has been accomplished recently by British scientists at the University of Bristol led by Julian Eastoe. How did they achieve this wonderful task? They did this by dissolving iron in a range of different surfactants. The surfactants used are not even novel but rather just ones you find in everyday materials like mouthwash and fabric softener. The addition of the iron creates metallic centers within the soap particles (as a biologist I am thinking something like iron in attaching to the heme groups).
However, when they tested it magic seemed to happen. They took a magnet to the test tube containing the soap and a less dense organic layer (probably some kind of oil). So oil on top soap on the bottom. When the magnet was introduced the iron-rich soap overcame gravity, as well as the interfacial tension between the water and the oil. The right image above shows the oil being disrupted in this case.
This research is interesting and has several implications in science and research and ecology. They report that the soap's electrical conductivity, surface tension, melting point, size and shape of the aggregates and how it dissolves in water are altered by the magnet which acts a simple on/off switch when introduced. The use of this on/off switch might make these kind of surfactants used only when it is needed and could potentially be removed after they are used. It is noted that this could help clean up oil spills better like the one that British Petroleum helped cause a couple of years ago in the Gulf of Mexico.
Taken originally from TG Daily Jan 24th, 2012.
Extra note: One has to think whether a few pints did not help them think about adding iron to soap. Alcohol helps science once again.
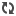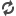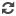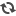 Logging you in...
Logging you in...