'Dyne With Me' explores surface tension of solutions used in everyday life. From inks to alcohol, drugs to dish soap, and everything in between....
Thursday, 4 August 2011
One reason to watch Nascar
In the previous post this group from Harvard claims they can use a chip with some wetting agent to detect the presence of methanol in bootleg (moonshine) solutions. This might be possibly using this agent. However it is not commercially available so I was thinking if it is possible to do with a normal tensiometer.
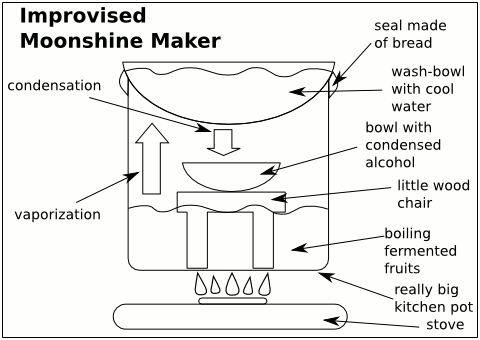
I am not making my own liquor or anything like that. Bootleg liquor (which got its name from originally came from concealing hip flasks of alcohol in the legs of boots) is a problem in small towns in Finland. During prohibition in the US bootlegged liquor made its way using fast stockcars paving the way for the racing sport: NASCAR. Finland unfortunately does not have that tradition of racing away from the police to deliver quality whiskey. However, in Finland in small towns if you see bottles on the side of the road you can stop, honk your horn and someone will come by with some moonshine or pontikka as it is called here. But one should be careful as it is quite easy for there to be quite a high content of methanol in this drink. The methanol vaporizes at a lower temperature than ethanol. So it if it not cooked at a high enough temperature or the people are making it without concern for purity they will leave the first few drips from the 'foreshot' in the moonshine.
People can test this in a couple different ways. The distiller may test this by putting it in a spoon lighting it and seeing if there is a blue flame (methanol burns with an invisible flame). This is not the best method. There is also a difference in mass transfer due to the Maragoni effect and by dropping some of the moonshine in a glass you might be able to see this compared to the same proof of alcohol. However, to test this a good method would be to use surface tension somehow. Although I have not figured out this when I wrote this. If someone knows let me know. I did do a brief review of the literature to find some interesting papers. One can also consult this paper using tensiometers or this paper using Monte Carlo simulations to see different ways to test the surface tension of methanol and ethanol water solutions. However it does not test methanol in ethanol water solutions. Using HPLC this paper derivatizates methanol to find the content in ethanol water solutions. It has excellent precision, accuracy and both intraday and interday reproducibility . It can also do large batches. However, it is not so portable and probably costs a lot for this.
If anyone knows how to measure the quantity of methanol in an ethanol-water solution I would love an answer.....
Labels:
drinking,
drinking surface tension,
ethanol,
methanol,
moonshine
Comments by IntenseDebate
Posting anonymously.
One reason to watch Nascar
2011-08-04T03:29:00-07:00
Unknown
drinking|drinking surface tension|ethanol|methanol|moonshine|
Subscribe to:
Post Comments (Atom)
 Logging you in...
Logging you in...